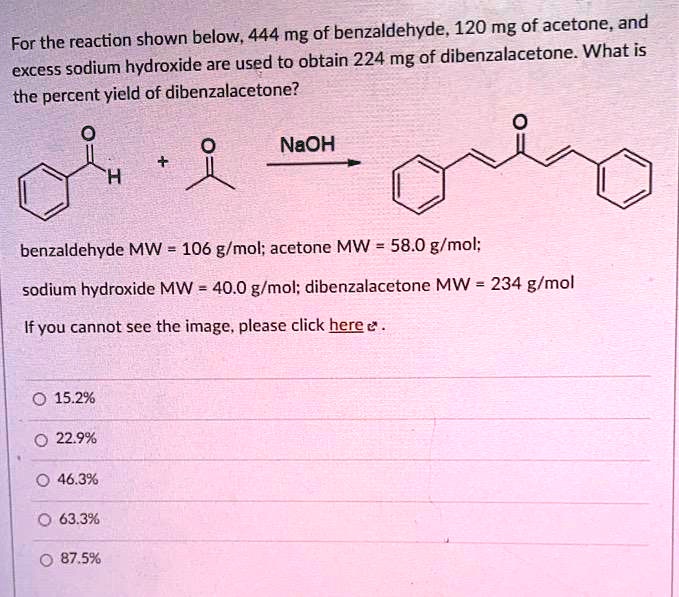
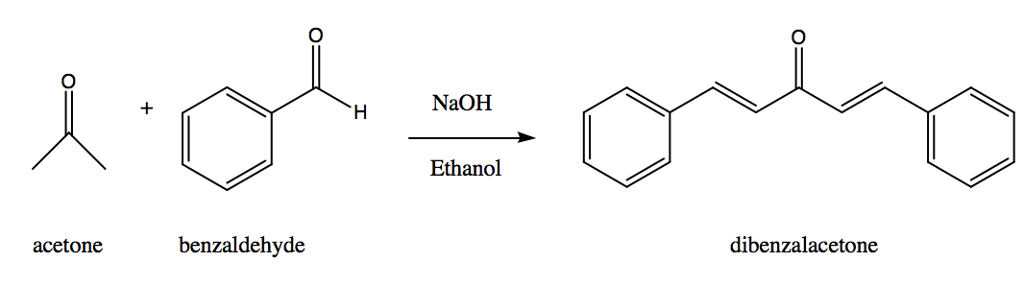
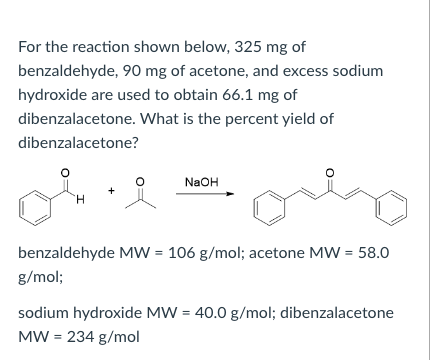
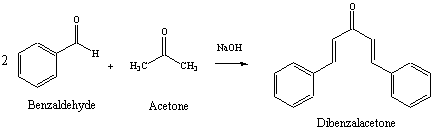
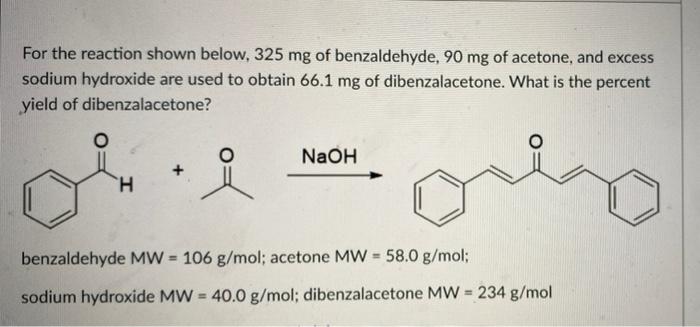
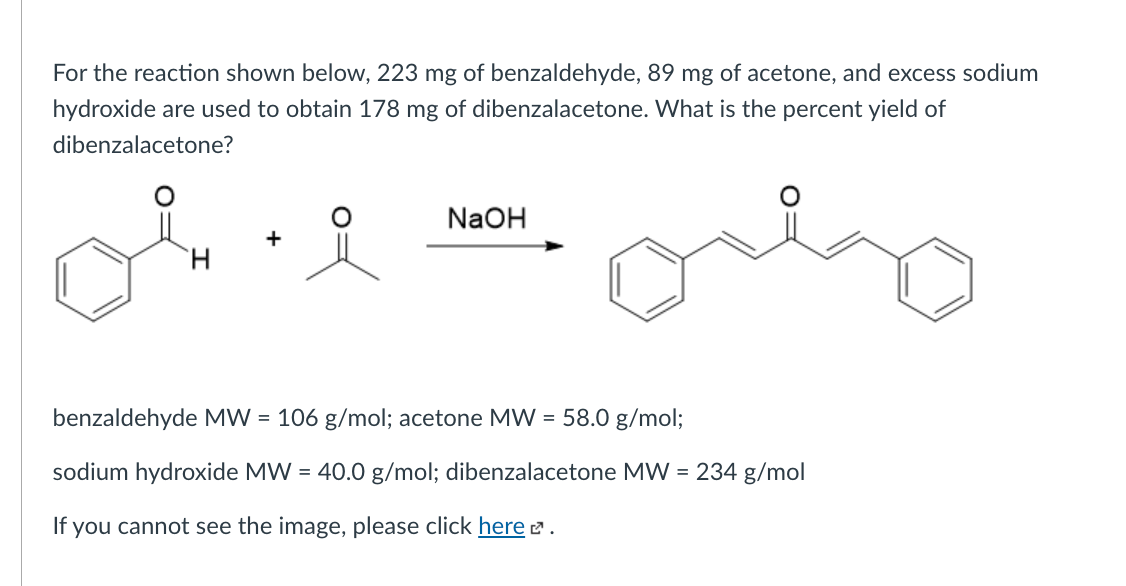
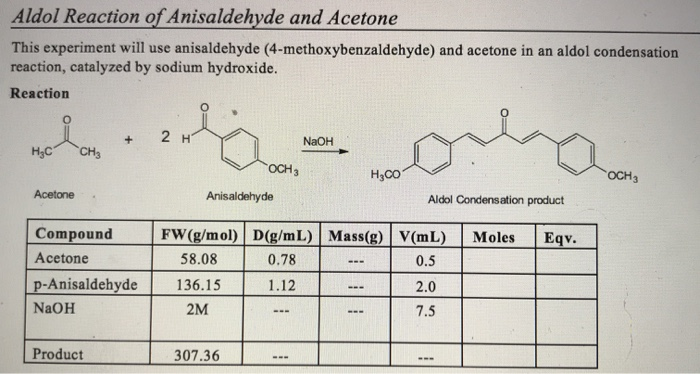
The product formed by aldol condensation between benzaldehyde and acetone is benzalacetone. Its structure is:

Mix 4.0 mL of benzaldehyde with 1 mL of acetone in 26 mL of 10% sodium hydroxide solution. (Density of benzaldehyde is 1.04 g/mL, density of acetone is 0.79 g/mL and density

SOLVED: 2 Why are you adding the mixture of acetone benzaldehyde dropwise to the sodium hydroxide solution? Why not add the acetone to the sodium hydroxide solution and then add the benzaldehyde

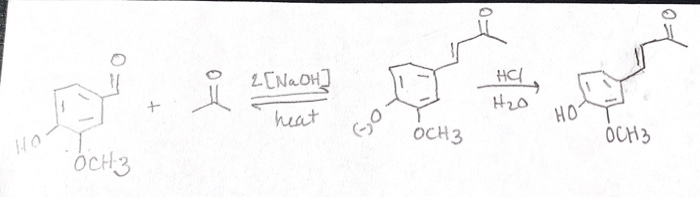
What reactions result from the addition of sodium hydroxide to the mixture of acetone and vanillin? | Homework.Study.com
Sciencemadness Discussion Board - What did I make? (Experiment w/ Acetone, Sulfur, and NaOH) - Powered by XMB 1.9.11
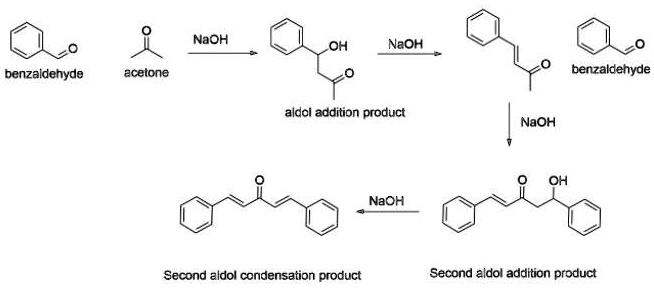
SOLVED: NaOH OH NaOH benzaldehyde acetone benzaldehyde aldol addition product NaOH OH NaOH Second aldol condensation product Second aldol addition product