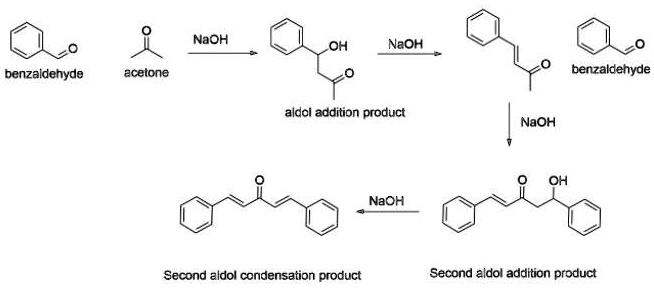
SOLVED: NaOH OH NaOH benzaldehyde acetone benzaldehyde aldol addition product NaOH OH NaOH Second aldol condensation product Second aldol addition product
Scheme 1 Mannich reaction between benzaldehyde 1, aniline 2 and acetone 3. | Download Scientific Diagram

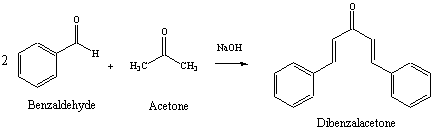
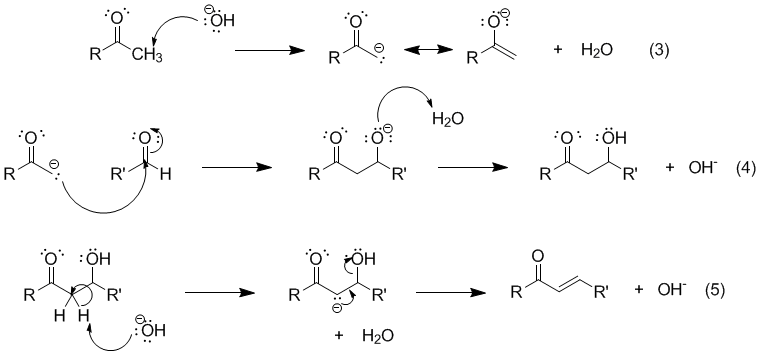
Give a detailed mechanism for the reaction (show all steps from acetone plus benzaldehyde to dibenzylacetone). | Homework.Study.com
Scheme 3: Aldol condensation of benzaldehyde and acetone followed by... | Download Scientific Diagram

Benzylidene acetone is the product of mixed condensation between benzaldehyde and acetone. What is its structure?

73. 2) Clemmenson reduction Benzaldehyde and acetone in 2:1 molar ratio is treated with base Ba(OH)2 as follows 4) NaBH 2 benzaldehyde +acetone #0 → Product is OH" 1) 3) Ph H 4) OO 74. PA Ph Ph. Ph CHO Ph
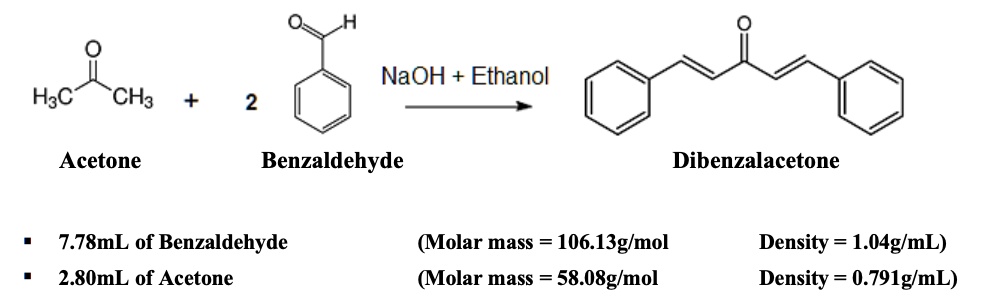
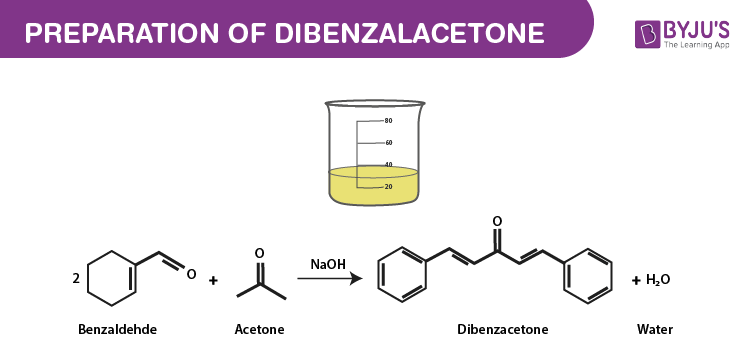
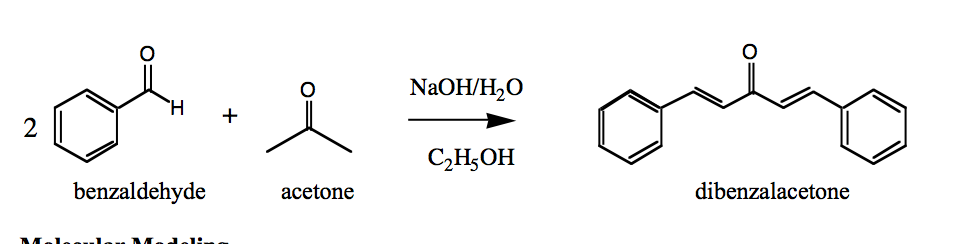
SOLVED: NaOH Ethanol HCl Acetone Benzaldehyde Dibenzalacetone 7.78 mL of Benzaldehyde 2.80 mL of Acetone (Molar mass = 106.13 g/mol) (Molar mass = 58.08 g/mol) Density = 1.04 g/mL Density = 0.791 g/mL CH3

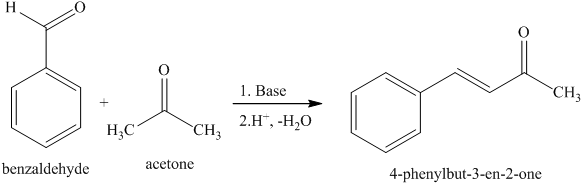
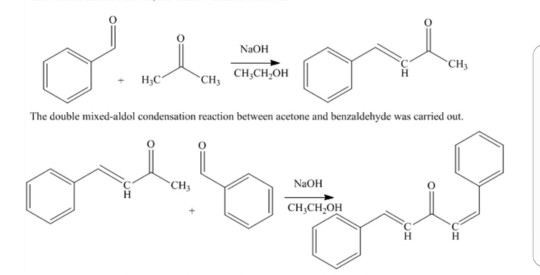
Write out all the steps in the mechanism for the base catalyzed condensation of acetone with 2 mol of benzaldehyde. | Homework.Study.com
![PDF] Reaction Efficiency of Crossed-Aldol Condensation between Acetone and Benzaldehyde over ZrO2 and ZrO2-Montmorillonite Catalyst | Semantic Scholar PDF] Reaction Efficiency of Crossed-Aldol Condensation between Acetone and Benzaldehyde over ZrO2 and ZrO2-Montmorillonite Catalyst | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0a54e47c87151b8af017126cb18d23eebb7b47e5/4-Figure4-1.png)
PDF] Reaction Efficiency of Crossed-Aldol Condensation between Acetone and Benzaldehyde over ZrO2 and ZrO2-Montmorillonite Catalyst | Semantic Scholar

Benzylidene acetone is the product of mixed condensation between benzaldehyde and acetone. What is its structure?