SOLVED: What mass of acetone (C3H6O) vapor would fill a 250-mL flask at a temperature of 100°C and a pressure of 760 torr? Group of answer choices: 2.11 grams 1.77 grams 3.99
![ratu, calculate the molar mass of the following compounds. urea (CO(NH2),] 9 mol acetone [CH,COCH3] Book iii) boric acid [H,BO3] iv) sulphuric acid [H 302] ratu, calculate the molar mass of the following compounds. urea (CO(NH2),] 9 mol acetone [CH,COCH3] Book iii) boric acid [H,BO3] iv) sulphuric acid [H 302]](https://toppr-doubts-media.s3.amazonaws.com/images/2005056/761575ee-b865-43ad-a8fd-1bd55e37e22a.jpg)
ratu, calculate the molar mass of the following compounds. urea (CO(NH2),] 9 mol acetone [CH,COCH3] Book iii) boric acid [H,BO3] iv) sulphuric acid [H 302]
![ratu, calculate the molar mass of the following compounds. urea (CO(NH2),] 9 mol acetone [CH,COCH3] Book iii) boric acid [H,BO3] iv) sulphuric acid [H 302] ratu, calculate the molar mass of the following compounds. urea (CO(NH2),] 9 mol acetone [CH,COCH3] Book iii) boric acid [H,BO3] iv) sulphuric acid [H 302]](https://toppr-doubts-media.s3.amazonaws.com/images/4457608/2f1ef40f-b4d0-4f2a-be92-bcdbf91493db.jpg)
ratu, calculate the molar mass of the following compounds. urea (CO(NH2),] 9 mol acetone [CH,COCH3] Book iii) boric acid [H,BO3] iv) sulphuric acid [H 302]
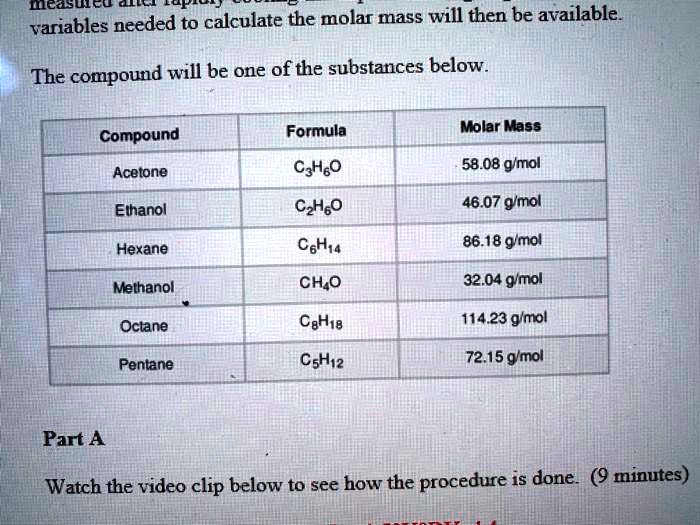
SOLVED: Molecules 4 variables needed to calculate the molar mass will then be available: The compound will be one of the substances below Formula Molar Mass Compound Acetone C3H6O 58.08 g/mol Ethanol

SOLVED: What is the mass (in g) of 1.935 mol of acetone, CH3COCH3? The molar mass of acetone is 58.08 g*mol-1.

SOLVED: Calculate the mass of acetone, C3H6O, that vaporizes as a result of absorbing 500 kJ of energy. The molar enthalpy of vaporization of acetone is 31.3 kJ/mol.

A student determines the molar mass of acetone, CH,COCH, by the method used in m found that the equilibrium temperature of a mixture of ice and water was 1.0°C on of a
![Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube](https://i.ytimg.com/vi/CCWl-z3Wdmo/sddefault.jpg)
Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube

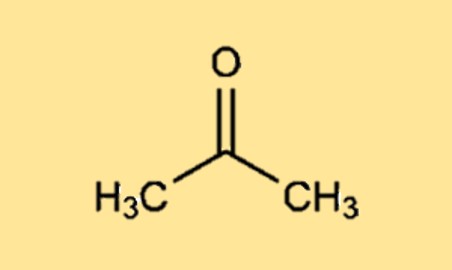
Acetone, or propanone, is an organic compound with the formula (CH3)2CO. It is the simplest and smallest ketone. It is … | Chemical structure, Chemistry, Molar mass







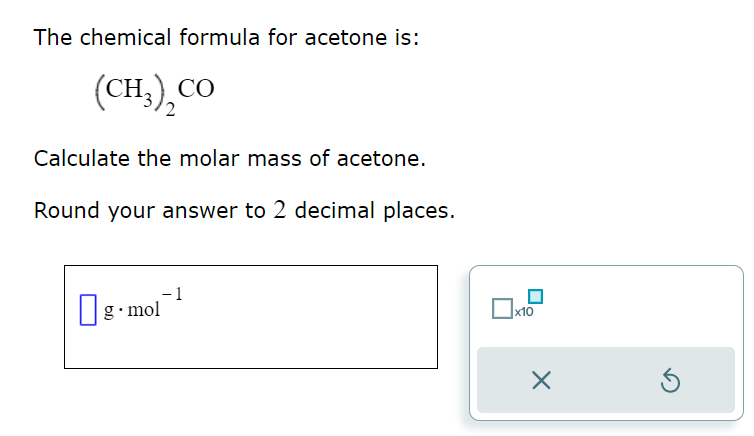
![Acetone [(CH3)2CO] Molecular Weight Calculation - Laboratory Notes Acetone [(CH3)2CO] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/02/acetone-molecular-weight-calculation-300x200.jpg)